Scientists at Institute for Systems Biology (ISB), University of Luxembourg, and Tampere University of Technology have created a method that identifies the genetic toggle switches that determine a cell’s developmental fate. This research, published on April 21 in the journal Nature Methods, may lead to new discoveries in disease treatments and tissue-regeneration technologies.
“In this elegant work, the authors propose a new way to identify genetic factors that influence cell fate based on the analysis of gene regulatory networks,” said Paul Brazhnik, PhD, of the National Institutes of Health’s National Institute of General Medical Sciences, which partly funded the work. “This new approach will advance our understanding of how the genome unfolds during an organism’s development and may provide a methodological framework for rational cell fate reprogramming for research and therapeutic purposes.”
There are more than 250 types of human cells, such as blood, skin and nerve, that develop from embryonic stem cells in a process called differentiation. The mechanisms controlling cellular differentiation have remained poorly characterized until now. This new paper (“Gene-pair expression signatures reveal lineage control”) presents a global picture of the regulatory circuitry behind the process – a family tree that maps the relationships between cell types – and reveals how cells can be reprogrammed.
Cell differentiation was imagined by British biologist Conrad Waddington in the 1950s as a journey from a peak of a hill down valleys and canyons of “cell fates” where mature cells ultimately take their final form. As the cell descends into each canyon, or pathway, it further becomes specialized. While this “epigenetic landscape” metaphor is vivid, it offers no practical insight into what determines the route or fate of the differentiating cell.
“The landscape concept is increasingly recognized to be central to much of biology, however” explained Sui Huang, PhD, a faculty member at Seattle-based ISB and one of the authors of the paper. “It is a tool to translate genes into phenotype.”
Huang has spent the past decade reviving Waddington’s theory, demonstrating its connection to co-author Stuart Kauffman’s theories of gene networks and publishing seminal papers that contributed to the basis of this current work.
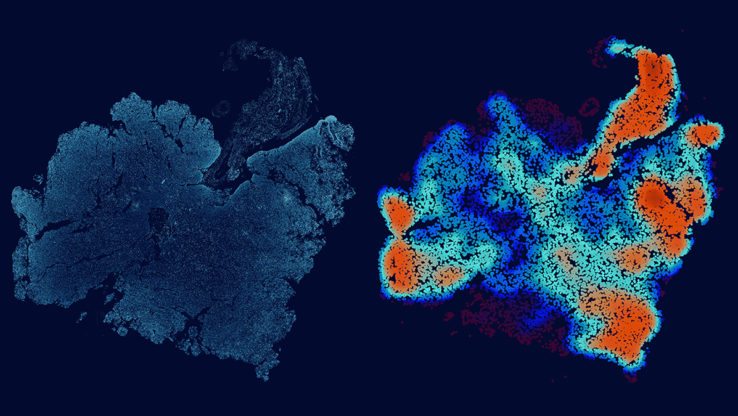
The researchers developed a novel computational approach for examining existing gene expression data to discover gene pairs that could participate in expression reversals – the toggle switch. The expression reversal is a signature of that pair of genes participating in a decision between two sister lineages. Prior to differentiating, the activity of both regulatory genes is equivalent. However as one regulatory gene increases, the opposing regulatory gene in the pair decreases, and results in cell differentiation. Flip the ratio of the pair and you now have a sister cell type. The team manually curated 166 different cell types and validated gene pairs that were previously implicated in cell differentiation, specifically blood stem cells and their differentiation into white or red blood cells. See figure below.
 “Waddington was perhaps one of the earliest system biologists,” said Ilya Shmulevich, PhD, ISB faculty member and co-author. “With a new computational approach, we were able to reconstruct his epigenetic landscape directly from the data.”
“Waddington was perhaps one of the earliest system biologists,” said Ilya Shmulevich, PhD, ISB faculty member and co-author. “With a new computational approach, we were able to reconstruct his epigenetic landscape directly from the data.”
Shmulevich added that there are many new pairs of regulatory genes that the analysis produces that were never considered. Providing the community with a resource to identify new factors for cell reprogramming and new hypotheses that could be tested in the lab is one of the values of this research.
“With this method, we will be able to estimate the height of the hill to be surmounted when making the cell jump from one valley to another,” said Shmulevich. “In the future, you could turn skin cells into heart muscle cells. The idea is that you wouldn’t need embryonic stem cells to do this. You could use your own skin cells to do it.”
Related Content:
- The research is online at http://trel.isbscience.net and a manual for interacting with the online tool can be found here. Waddington’s formerly metaphorical Epigenetic Landscape, now formed by concrete data, can be viewed here.
- The paper was the result of three years of collaborative work by ISB, Luxembourg Centre for Systems Biomedicine at the University of Luxembourg, and Stuart Kauffman, a pioneer in the field of complex systems science and an affiliate faculty member of ISB. The study was supported in part with funds from the Grand Duchy of Luxembourg and the National Institutes of Health grants GM072855 and GM076547.



 isbscience.org/research/biggest-family-tree-of-human-cells-may-help-develop-cell-replacement-therapies/
isbscience.org/research/biggest-family-tree-of-human-cells-may-help-develop-cell-replacement-therapies/